Fission Definition and 311 Threads
-
J
B Why does Lithium fission produce energy?
Hi, a simple question from an amateur: Why does Lithium fission produce energy instead of consuming it? (https://www.world-nuclear.org/information-library/current-and-future-generation/lithium.aspx) I always thought that fission of heavy elements produces energy, and fusion of light elements...- Jackissimus
- Thread
- Fission Lithium
- Replies: 1
- Forum: High Energy, Nuclear, Particle Physics
-
S
Calculating Energy Released in a Fission Reaction: Where Did I Go Wrong?
Energy = ##\frac{2000}{235}## x 6.02 x 1023 x 0.7% x 200 x 1 x 106 x 1.6 x 10-19 = 1.15 x 1012 J But the answer is 1.15 x 109 J I am off by a factor of 1000. Where is my mistake? Thanks- songoku
- Thread
- Energy Fission Reaction
- Replies: 6
- Forum: Introductory Physics Homework Help
-

A Probability of U-235 fission happening
Where can I find information about the ~82% of U-235 nuclear fission happening and ~18% not happening?- NewPhysi
- Thread
- Fission Probability
- Replies: 3
- Forum: High Energy, Nuclear, Particle Physics
-

I Nuclear Fission of Uranium-235
I'm learning about nuclear fission for fun, well based on my research, 82% of the time that uranium-235 absorbs a neutron it will fission, the uranium-235 will briefly turn into an excited uranium-236 and after that, it will split, the other 18% of the time it will just emit radiation and...- NewPhysi
- Thread
- Fission Nuclear Nuclear fission Uranium
- Replies: 3
- Forum: High Energy, Nuclear, Particle Physics
-

Binding energy and nuclear fission
If I am not mistaken, binding energy is the energy required to separate the constituents of the nucleus, and is always a positive number. However, if splitting elementary particles in the nucleus apart requires energy, then why do fission reactions release energy?- Leo Liu
- Thread
- Binding energy Energy Fission Nuclear Nuclear fission
- Replies: 29
- Forum: Nuclear Engineering
-
S
When do fission products get cooler than fuel?
Main nuclear fuel is U-235. Actinium series to stable Pb-207 includes 7 alpha and 4 beta decays. Total energy yield 46,4 MeV. In the main branch, the 4 beta decays carry around 3 MeV energy. Meaning the alpha decays are about 43 MeV... and they have high biologic effectiveness, around 20...- snorkack
- Thread
- Cooler Fission Fuel
- Replies: 6
- Forum: Nuclear Engineering
-
M
Other Creating a Nuclear Fission Reactor
How do I create a nuclear fission reactor? if you can link me up to any places to buy the things or any other sites that would be good.- MatthewN
- Thread
- Fission Nuclear Nuclear fission Reactor
- Replies: 1
- Forum: STEM Academic Advising
-
A
Fission Products that come from the MCNP output?
What are the most important fission products should I include/care about that comes out from the MCNP output?- Aly_19f
- Thread
- Fission Mcnp Output
- Replies: 3
- Forum: Nuclear Engineering
-
H
Electrons and their little role in nuclear physics
In this thread, @haruspex presented a very deliberate point about the role of electrons in a nuclear fission reaction (he might have said or meant something else but I will present my version of it). The problem that we have before us can be stated, as candidly as my linguistic faculty of mind...- Hall
- Thread
- Electrons Fission Nuclear Nuclear physics Physics
- Replies: 1
- Forum: Introductory Physics Homework Help
-

Finding the distance between two nuclei right after fission
I am trying to solve part b of this problem. I know the particles are moving at first and are at rest right after fission per the question. Using the relativistic Kinetic Energy equation and the energy between two charges I believe I can solve for r, the radius between the two charges. I am a...- guyvsdcsniper
- Thread
- Fission Nuclei
- Replies: 13
- Forum: Introductory Physics Homework Help
-
J
B Nuclear Fission & Creation of Plutonium
Just joined the forum after youtubes algorithm suggested a story documenting the ‘Chicago Pile’. I ended up watching a bunch of other power plant videos becoming more confused with each one. I apologize up front as I know nothing about the field of nuclear physics and not even sure if I’ll even...- JLynch
- Thread
- Creation Fission Nuclear Nuclear fission
- Replies: 8
- Forum: Classical Physics
-

Halite / Centurion and fission - fusion power plants
Its unclear to me what exactly these tests entailed and how much today is still top secret. They involved inertial confinement fusion with x-rays from a fission explosion. never see this discussed - forgetting about political issues with fission, how feasible is a fusion reactor utilizing...- BWV
- Thread
- Fission Fusion Fusion power Plants Power
- Replies: 7
- Forum: Nuclear Engineering
-
L
Calculation on spontaneous fission and effect from alpha decay
Summary:: Help needed on how to calculate on spontaneous fission and effect from alpha decay Heat from alpha decay from Pu-238 is used to generate direct current. At the beginning (1977) it generated 470W, how large is the effect now? And if the efficiency between the electricity and heat...- Luxdot
- Thread
- Alpha Alpha decay Calculation Decay Fission Spontaneous
- Replies: 23
- Forum: Introductory Physics Homework Help
-
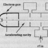
More info about natural nuclear fission going critical 2 Million years ago?
This week I read through the chapter "Energy from the Nucleus" inside the familiar intro physics textbook Halliday, Resnick, Walker. I found it fascinating that there is evidence from a uranium mine in West Africa that there may likely have been a nuclear fission natural reactor that went...- CPW
- Thread
- Fission Natural Nuclear Nuclear fission Years
- Replies: 4
- Forum: Earth Sciences
-
S
I Fission vs Cluster Decay: What is the Difference?
What is defined as "fission"? Isotopes that undergo "fission" under influence of fast neutrons, though not slow neutrons, are classified as "fissionable". Is Be-9 fissionable? What is the dividing line between "fission" and "cluster decay"?- snorkack
- Thread
- Fission
- Replies: 4
- Forum: High Energy, Nuclear, Particle Physics
-
A
Fusion reactors are neutron sources AKA fission bomb factories?
Assumptions: 1) Fusion reactors are easily obtainable. Every spacecraft or hover-car has one. 2) Fusion reactors are neutron sources. 3) Neutron sources are the "difficult bit" in the enrichment of uranium and plutonium. 4) Uranium and plutonium are "freely" available in asteroids or...- ardnog
- Thread
- Bomb Fission Fusion Neutron Sources
- Replies: 7
- Forum: Science Fiction and Fantasy Media
-
T
MCNP 6.2: Fission Product Recoil/Transport
I am trying to simulate fission product ejection from thin fissile films in gas filled detectors (fission chamber). Does MCNP 6.2 produce recoil fission products that will be transported through the system? I have enabled "heavy ion physics" (#), tried options 3 and 5 for NCIA on the neutron...- TheNE
- Thread
- Fission Mcnp Product
- Replies: 3
- Forum: Nuclear Engineering
-
F
Is MCNP capable of scoring fission spectrum?
Hi everybody, I am trying to score fission spectrum in MCNP for a kcode calculation. I would like to check at which energy neutrons produced by fission are generated. I have no idea how to perform since tallies are usually volume or material dependent and I just want to build a spectrum...- froztiz
- Thread
- Fission Mcnp Spectrum
- Replies: 2
- Forum: Nuclear Engineering
-

Relativistic Mass in this fission decay
I'm not sure where to start- mtuck
- Thread
- Decay Fission Mass Relativistic Relativistic mass
- Replies: 3
- Forum: Introductory Physics Homework Help
-
S
How many neutrons are produced in nuclear fission?
The nuclear fission reaction is: ##U_{92}^{235} + n^{1}_{0} \to Ba_{56}^{141} +Kr_{36}^{92} + 3 ~n^{1}_{0} ## I am not sure about the number of neutrons produced. Is it 2 or 3? 2 is from the number of neutrons on the RHS - number of neutron on LHS 3 is from the number of neutrons on RHS only...- songoku
- Thread
- Fission Neutrons Nuclear Nuclear fission produced
- Replies: 9
- Forum: Introductory Physics Homework Help
-
T
Nuclear fission calculation of energy released
Summary:: Calculate the amount of energy in joules generated from 2 kg of uranium fuel if the uranium 235 represents 0.7% of the metal and every fission releases 200 MeV. Hi! I am stuck in this question from my exercise book : Q. Calculate the amount of energy in joules generated from 2 kg...- tahmidbro
- Thread
- Calculation Energy Energy released Fission Nuclear Nuclear fission
- Replies: 9
- Forum: Introductory Physics Homework Help
-
A
Principle of Nuclear Fission Direct Energy Conversion
Good day everyone, I've recently been researching direct energy conversion schemes for nuclear fission, and I have a question on the basic physics behind the device (specifically on how it converts kinetic energy to electricity). In essence, the "basic" scheme for fission DEC devices is that...- Aakash Sunkari
- Thread
- Energy Energy conversion Fission Nuclear Nuclear fission Principle
- Replies: 5
- Forum: Nuclear Engineering
-

I The surface area of an oblate ellipsoid
In "An Introduction to Nuclear Physics by W. N. Cottingham, D. A. Greenwood" for the surface area of an oblate ellipsoid, the following equation is written for small values of ε : The book has said this without proof. I found the following formula for the desired shape: No matter how hard I... -
B
I Questions about the energy released by nuclear fission
as far as i understand nuclear fission is the splitting of a heavy nucleus cause of a added neutron the energy you get is cause of the lost mass from binding energy. but i also read that the nuclear fission products undergo beta decay which emits beta particles gamma rays and antineutrinos so...- brent1369
- Thread
- Energy Energy released Fission Nuclear Nuclear fission
- Replies: 4
- Forum: High Energy, Nuclear, Particle Physics
-
A
Can a Fission Fragment rocket be designed without a moderator?
a dusty plasma design : https://www.nasa.gov/pdf/718391main_Werka_2011_PhI_FFRE.pdf has nuclear fuel held in place by electromagnetic fields. It uses a very massive moderator around the fuel to slow and reflect neutrons back to the fuel, to enable continued fission. But, there is one kind of...- Al_
- Thread
- Fission Moderator Rocket
- Replies: 20
- Forum: Nuclear Engineering
-
B
I What is the Minimum Neutron Energy Required for Lithium Fission?
Whats the minimum kinetic energy a neutron must have in order to trigger the fission of for example a lithium core ?- B4cklfip
- Thread
- Core Energy Fission Neutron
- Replies: 1
- Forum: High Energy, Nuclear, Particle Physics
-
P
Fission products released from failed fuel rods in a PWR
There are a lot of fission products accumulated in the fuel pellet and gap in the fuel cladding during PWR operation. Howeve, some iodine and noble gas will release to the primary loops in case of fuel failure. The fission product release rate can be described as the escape rate coefficent which...- Pengtaofu
- Thread
- Fission Fuel
- Replies: 7
- Forum: Nuclear Engineering
-

Fission vs. Fusion question (particle physics)
Homework Statement: Calculate energy released in the reactions below, and compare them (see full question below) Homework Equations: a) FISSION: 1g of U-235 splits into La-148 + Br-87 b) FUSION: 0.5g of D2O with 0.5g of T20 creates He-4 and a neutron useful equations: E = Δm(c^2) Hey, can...- Diyon335
- Thread
- Fission Fusion Particle physics Physics
- Replies: 4
- Forum: Introductory Physics Homework Help
-
F
I Fissionable elements for Fission
Hello, Quick question: in fission, which is a nuclear reaction, a large element is hit with neutrons and broken/split into two smaller elements while lots of energy is released. Does the starting element, which must be large in size (like uranium-235 or plutonium-239) need to be an element that...- fog37
- Thread
- Elements Fission
- Replies: 2
- Forum: High Energy, Nuclear, Particle Physics
-

I What Natural Phenomena Resemble Nuclear Fission?
Is there any phenomenon in nature that resembles or is analogous to nuclear fission?- Ranku
- Thread
- Fission Natural Nuclear Nuclear fission Phenomena
- Replies: 5
- Forum: High Energy, Nuclear, Particle Physics
-
A
B Analysis of Fusion & Fission by Binding Energy
In fusion reactions, lighter nuclei are combined into heavier ones (atomic mass increases); so the direction is from left to right (shown on the diagram). Therefore, in the fusion of 'light elements' the total final binding energy is higher (the related arrow points upward); so ##B_2-B_1>0## and...- A M
- Thread
- Analysis Binding energy Energy Fission Fusion
- Replies: 3
- Forum: High Energy, Nuclear, Particle Physics
-
A
B Basic definition: Nuclear Fusion & Fission
The best-known classes of nuclear transmutations are fission and fusion: Nuclear Fusion Under normal condition, nuclei do not stick together; because they repel each other at large distances (due to the electrostatic repulsion 'barrier') and thus the strong nuclear force cannot act. But if these...- A M
- Thread
- Definition Fission Fusion Nuclear Nuclear fusion
- Replies: 5
- Forum: High Energy, Nuclear, Particle Physics
-

MCNP4c2: Fission Reactions in a Spherical Subcritical Reactor
Hi, i am new to simulation and for my thesis i have to make a simple simulation by using mcnp4c2. Is anybody familiar with this version of MCNP? I need to calculate the fission reactions per second in a geometry of a spherical sub critical reactor of Uranium with low percentage of U 235 with...- Dimitris Catzis
- Thread
- Fission Reactions Reactor Spherical
- Replies: 3
- Forum: Nuclear Engineering
-
J
Can Someone Explain How Nuclear Fuel Rods Heat Water in a Reactor?
Hi, I haven't studied much nuclear physics and would be very grateful if someone could answer a few basic questions I had about fission in the fuel rods on how the water is actually heated in the reactor core: 1) I don't know if I have this correct but is it that the way the water is heated in...- Jimmy87
- Thread
- Fission Fuel Nuclear Nuclear fuel
- Replies: 10
- Forum: Nuclear Engineering
-
C
Safe zero base reactivity level nuclear fission reactor?
There are already 4 generations of nuclear fission reactors. (Compare https://en.wikipedia.org/wiki/Nuclear_reactor#By_generation.) However among these, there does not seem to be a (maximum) safe nuclear fission reactor design, which immediately stops the nuclear chain reaction when the reactor...- consuli
- Thread
- Base Fission Nuclear Nuclear fission Reactor Reactor design Zero
- Replies: 2
- Forum: Nuclear Engineering
-
V
I Binding energy, fusion and fission
Hi. My high school physics book doesn't elaborate the idea of binding energy and how it's related to fissions and fuisions adequately in a way that made me have wrong thoughts about these ideas. What i understand after doing some research is that: - Binding energy is the energy that has to be...- Volta
- Thread
- Binding energy Energy Fission Fusion
- Replies: 3
- Forum: High Energy, Nuclear, Particle Physics
-
C
B Energy conservation in nuclear fission
Hi all, I struggle to understand how energy is conserved I fission. If the binding energy per nucleon increases, surely the mass defect simply accounts for that difference to conserve energy before and after. How does the mass defect account for the kinetic energy of the fission fragments as well?- cormzy
- Thread
- Conservation Energy Energy conservation Fission Nuclear Nuclear fission
- Replies: 1
- Forum: High Energy, Nuclear, Particle Physics
-
C
I Why is Proton Radiation this Rare in Nuclear Fission Decay?
The atomic nuclei consist out electrons, protons and neutrons (with only exception of hydrogen, that does have a neutron). Thus, it would be straightforward, that there existed a corresponding radiation for each nucleus component, when a nucleus decays. There is an electron radiation (beta...- consuli
- Thread
- Alpha radiation Decay Fission Nuclear Nuclear fission Particle Proton Radiation
- Replies: 9
- Forum: High Energy, Nuclear, Particle Physics
-

I How much energy does a fission nuke actually release?
So I was trying to debunk a conspiracy theory involving bombs and I realized that I have a small problem with Einstein's equation: E=mc2 To be honest I haven't really understood how it's used... I mean, how far should the fission go until as much energy as E=mc2 is released? Does the matter have...- Mohammad Hunter
- Thread
- Energy Fission Nuke Release
- Replies: 4
- Forum: High Energy, Nuclear, Particle Physics
-
J
B Terrestrial Spontaneous Fission & Unstable Isotopes
Does terrestrial spontaneous fission or natural fission create unstable isotopes similar to nuclear fission in reactors?- Jorge cunha
- Thread
- Fission Isotopes Spontaneous
- Replies: 1
- Forum: High Energy, Nuclear, Particle Physics
-

Neutron capture and fission reaction
Hi, I've been wondering about this a lot and I didn't find a satisfactory answer by myself. My professor said that in order to promote a fission reaction you need to provide a certain amount of energy (like in a chemical reaction you need to overcome the activation energy) and the easiest way...- dRic2
- Thread
- Capture Fission Neutron Neutron capture Reaction
- Replies: 15
- Forum: Nuclear Engineering
-
S
I Could a Smoke Detector Serve as a Sail for Spacecraft Propulsion?
Looking at a humble smoke detector - a metal plate with some alpha source on one side. I guess the fission products / apha etc emitted in one direction are absorbed into the metal, and the opposite direction go into space. So there should be a minute net force ? Would that be useful as a "sail"...- synch
- Thread
- Fission Radioactivity Rocket Source Space craft
- Replies: 2
- Forum: Other Physics Topics
-

B Where does the increase in mass come from in fission?
If energy is released during nuclear fission, why is the mass of the products (the two new nuclei and fission neutrons) greater than the mass of the original nuclei? In accordance to E=mc^2, shouldn't the release of energy result in the products having a lesser mass than the original nuclei?- Ciaran H
- Thread
- E=mc2 Fission increase Mass Nuclear
- Replies: 1
- Forum: High Energy, Nuclear, Particle Physics
-
A
B How energy is released during nuclear fission?
In a simple nuclear fission reaction an uranium atom breaks into krypton and barium releasing around 200MeV. Binding energy per nucleon (BE/A) of uranium = 7.6 MeV And binding energy per nucleon of krypton an barium is just larger than that of uranium. So in my view, around 200 MeV is used to...- aayush
- Thread
- Energy Fission Nuclear Nuclear fission
- Replies: 4
- Forum: High Energy, Nuclear, Particle Physics
-
T
Nulcear fission, two daughter nuclei
Homework Statement An unstable nucleus with mass M is in an excited state with excitation energy E* and undergoes spontaneous fission into two daughter nuclei "1" and "2" and zero fast neutrons. What will be the kinetic energy KE (in MeV) of the daughter nucleus "1"? M = 233.9493amu E* =...- Taylor_1989
- Thread
- Fission Nuclei
- Replies: 5
- Forum: Introductory Physics Homework Help
-

Inventory of all fission products for 50% fission of 1g U235
I would like to learn the initial quantities of all fission products for a 50% fission (nuclear yield) of 1g of U235. I roughly understand that that there could be over 200 fission products produced upon nuclear fission of U235. Is there any tools available for the above estimation ? Learned...- Wallace Chan
- Thread
- Fission
- Replies: 1
- Forum: Nuclear Engineering
-
D
Exploring Nuclear Power Technology: A Scientific Perspective
Hello. My name is Dom Napolitano and I would like to create a thread on Nuclear Power Technology.- Dominic Napolitano
- Thread
- fission fusion nuclear power nuclear reactors
- Replies: 2
- Forum: New Member Introductions
-

Could Induced Fission Be Achieved by Combining Proton and Electron Beams?
If you set up a proton beam, intersected the proton beam with an electron beam, and made sure the electron beam (or possibly the proton beam) was high enough energy to compensate for the missing electron anti-neutrino, could you produce a neutron beam? Could the neutron beam then be used to...- jlefevre76
- Thread
- Fission Induced
- Replies: 5
- Forum: Nuclear Engineering
-
K
B Why uranium dioxide is used in nuclear reactors?
In Wikipedia I read that Uranium-235 is a nuclear is fuel in fission reactors, also that Uranium-235 is split (fission) and energy is revealed. But in some images showing how nuclear reactors work, they show Uranium dioxide as nuclear fuel elements. In Wikipedia, it's written that the oxides...- Karagoz
- Thread
- Fission Nuclear Nuclear power plant Nuclear reactor Nuclear reactors Uranium
- Replies: 2
- Forum: High Energy, Nuclear, Particle Physics
-
V
B Simple doubt in nuclear fission
Hi all , I have a very simple doubt in the basics of radioactivity . Nuclear fission is the splitting of the nucleus of a heavy atom such as U23592 by bombarding with slow neutrons into two lighter nuclei with a simultaneous release of huge amount of energy . Now my doubt is that - Can a...- Vibhor
- Thread
- Doubt Fission Nuclear Nuclear fission
- Replies: 9
- Forum: High Energy, Nuclear, Particle Physics