- #1
gnurf
- 370
- 8
I recently came across MIT prof. Walter Lewin's fascinating demonstration of a Kelvin water dropper, which can be seen here:
https://www.youtube.com/watch?v=http://www.youtube.com/watch?v=oY1eyLEo8_A
(EDIT: If your browser is like mine that didn't work, try this: )
As shown in the diagram below, the setup consists of a bucket of water and some empty paint cans that act together in a positive feedback loop where the charge builds up to create a battery with a potential difference of several kilovolts. In the MIT demo, there are also two closely placed conductors from the each of the opposite charged buckets so that the buckets are discharged (resulting in a spark in the air between them) when the potential exceeds some threshold.
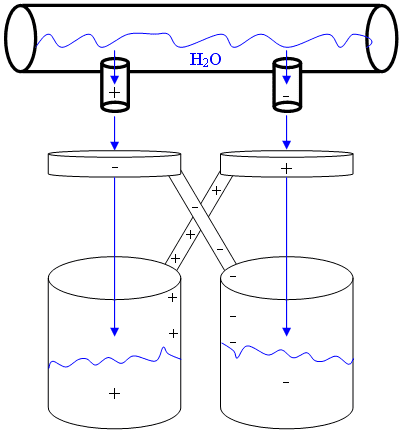
From wikipedia's http://en.wikipedia.org/wiki/Kelvin_water_dropper#Principle_of_operation" section:
Am I right to think that this wouldn't work with pure H2O (i.e an insulator with no free electrons)? Also, the prof. says that "the way it works is actually quite subtle", so I'm wondering the if the wikipedia entry is the whole story, or if there's more to it?
https://www.youtube.com/watch?v=http://www.youtube.com/watch?v=oY1eyLEo8_A
(EDIT: If your browser is like mine that didn't work, try this: )
As shown in the diagram below, the setup consists of a bucket of water and some empty paint cans that act together in a positive feedback loop where the charge builds up to create a battery with a potential difference of several kilovolts. In the MIT demo, there are also two closely placed conductors from the each of the opposite charged buckets so that the buckets are discharged (resulting in a spark in the air between them) when the potential exceeds some threshold.
From wikipedia's http://en.wikipedia.org/wiki/Kelvin_water_dropper#Principle_of_operation" section:
Any small charge on either of the two buckets suffices to begin the charging process. Suppose, therefore, that the left bucket has a small positive charge. Now the right ring also has some positive charge since it is connected to the bucket. The charge on the right ring will attract negative charge into the right-hand stream by electrostatic attraction. When a drop breaks off the end of the right-hand stream, the drop carries negative charge with it. When the negatively charged water drop falls into its bucket (the right one), it gives that bucket and the attached ring (the left one) a negative charge.
Am I right to think that this wouldn't work with pure H2O (i.e an insulator with no free electrons)? Also, the prof. says that "the way it works is actually quite subtle", so I'm wondering the if the wikipedia entry is the whole story, or if there's more to it?
Last edited by a moderator: