- #1
FishmanGeertz
- 190
- 0
In chemistry and biochemistry, what do the hexagons with letters in between them mean? Call me silly but for some reason they never taught this to us in school.
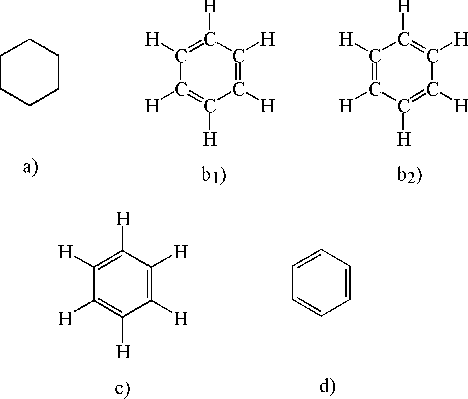
QuarkCharmer said:These shapes are called skeleton formulas. It's a short hand way of representing a molecule. Lines for single bonds, double lines for double bonds etc. It's assumed that each intersection point of the line segments is a molecule, usually carbon, but you can have structures that are not consisting of carbon atoms, these are called heteroatoms and you represent them by noting the symbol for the atom in the chain like this:
In your posted image, you would assume (since there is no notation) that each point is a carbon atom. Interestingly enough, the only way they can maintain that hex form is by having alternating double bonds, hence the alternating double lines in your image. Hope that helps. I am sure you can find more info now that you know what they are called.
In chemistry, hexagonal shapes often represent the structure of benzene rings or other aromatic compounds. These hexagonal structures signify the arrangement of carbon atoms in a ring, with alternating single and double bonds.
The benzene ring, a fundamental structure in organic chemistry, is represented as a hexagon because it consists of six carbon atoms connected in a ring formation with alternating double and single bonds. This hexagonal representation simplifies the depiction of its molecular structure.
Aromatic compounds are a class of compounds that include benzene as a part of their structure. These compounds are characterized by their stability and unique chemical properties, which are often represented in chemical diagrams as hexagonal structures.
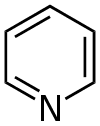
While hexagons in chemistry typically represent a ring of carbon atoms, the vertices of the hexagon can also include other elements like nitrogen in certain heterocyclic compounds. The hexagon primarily denotes the ring structure.
While the hexagonal shape is most famously associated with benzene, it is not unique to it. Other cyclic compounds with six members in their ring, including some non-aromatic compounds, can also be represented by hexagons.
The hexagonal representation of benzene reflects its delocalized bonding system, where electrons are shared across all six carbon atoms, contributing to its stability and unique chemical properties. The alternating single and double bonds are often depicted inside the hexagon.
Yes, hexagonal structures like benzene rings can be functionalized with various chemical groups. These groups can attach to the carbon atoms of the ring, altering the compound's chemical properties and reactivity.