Atom Definition and 999 Threads
-

Calculating potential energy of hydrogen atom
Homework Statement Calculate ##\left< \frac 1 r \right>## and ##\left< \frac 1 {r^2} \right>## and the expectation value and uncertainty of the potential energy of the electron and proton for a hydrogen atom in the given state. The given state is: $$ \psi_{2,1,-1} \left( r,\theta,\phi \right)...- Leechie
- Thread
- Atom Energy Hydrogen Hydrogen atom Potential Potential energy
- Replies: 4
- Forum: Advanced Physics Homework Help
-
C
I How can an atom change from ground to excited state?
I am currently studying a Foundation Program, which includes chemistry. -I can easily understand the distinguishable characteristics between ground and excited state via writing the electron configuration. -But how can an tom changes from ground to excited state ? And can an atom changes from...- ConfusedRookie
- Thread
- Atom Change Electron Electron configuration Energy change Excited Ground State
- Replies: 18
- Forum: Quantum Physics
-
M
I Why is spin-orbit coupling neglected in Helium atom?
I was looking at grotrian diagrams for helium and I see that there is no splitting of energy levels due to spin-orbit coupling. In my book it is said that spin-orbit coupling in helium is small and can be neglected but no further explanation is given. At the same time we do spin-spin coupling...- misko
- Thread
- Atom Coupling Helium Spin-orbit
- Replies: 5
- Forum: Atomic and Condensed Matter
-

I Equation for modeling atomic spectra of all atoms
I've seen the equation I think is just for hydrogen. is this just for hydrogen? of course this doesn't return the atomic spectra, it returns the energy. So using E=h*v and Planck's constant. a simple factor of 1/h would return the frequency. right? Energy is directly proportional to frequency...- Xilus
- Thread
- Atom Atomic Atomic spectra Atoms Modeling Spectra
- Replies: 6
- Forum: Atomic and Condensed Matter
-

Why Are Multiple Quantum Numbers Allowed in a Hydrogen Atom?
Homework Statement http://i.imgur.com/GQ9Xk6d.png Homework Equations Quantum mechanical model of atomic structure The Attempt at a Solution Why all sets are allowed? H atom only got one e- which only one orbital should be there, isn't it? If there aren't second or more e- , no second...- yecko
- Thread
- Atom Hydrogen Hydrogen atom Quantum Quantum number
- Replies: 2
- Forum: Biology and Chemistry Homework Help
-
G
I Predicting new crystal structures
Hello, Sorry if this is a rather basic question, but my memories of solid-state chemistry are a bit rusty. Basically I'm trying to predict new crystal structures. I understand the crystallographic aspects quite well and know that at given external temperature and pressure the most stable...- g_mogni
- Thread
- Atom Crystal Crystal lattice Crystal structure Structures
- Replies: 1
- Forum: Atomic and Condensed Matter
-
S
How Many Beams Are Hydrogen Atoms Deflected Into in a Magnetic Field?
Homework Statement A beam of neutral hydrogen atoms in their ground state is moving into the plane of the page and passes through a region of a strong inhomogeneous magnetic field that is directed upward in the plane of the page. After the beam passes through this field, in how many beams was...- Silviu
- Thread
- Atom Field Hydrogen Hydrogen atom Magnetic Magnetic field
- Replies: 12
- Forum: Advanced Physics Homework Help
-

B What particles make up an atom?
I'm trying to find a way to understand Q.P. So far I know that each element contains as many protons as it does nuetrons or electrons. Is that right? Derp lol- Hose way or joshua
- Thread
- Atom Particles
- Replies: 2
- Forum: High Energy, Nuclear, Particle Physics
-
E
B What Happens to Atom Properties When Electrons Are Removed?
hello everybody :) Let's assume that we could steal all electrons of an atom. What would happen? How would the atom change its properties? Could we measure different values?- entrance
- Thread
- Atom Atoms Electrons
- Replies: 16
- Forum: Atomic and Condensed Matter
-
G
A Paschen notation for electronic states of Argon atom
Hello. I think excited states of Argon atom is described by j,l coupling written as (2s+1)[K]J. However, when I read some papers describing cross section data for Argon atom, data are quoted with strange notation, Paschen notation. I tried to study the Paschen notation and found that its form...- goodphy
- Thread
- Argon Atom Electronic Notation Spectroscopic notation States
- Replies: 2
- Forum: Atomic and Condensed Matter
-

Nearest neighbour, nnn....etc. of an atom in a crystal
There are some software can provide visualization of crystal or protein structure, i.e. VESTA. I want to know how many atoms is the nearest neightbour (and their distance), and the next nearest neighbour...etc of a particular atom. Is there any software (or perhaps some data book?) can do this? -
L
How does the magnetization of an atom change at its center?
Homework Statement We have coaxial cabel with radius R and R0=3R filled with magnetic.Magnetization J(r)=(r3-R3)/(2R3)*I/(2*pi*r).What is the magnitization if r=0(center of atom)?And what a magnetization direction of atom? Homework EquationsThe Attempt at a Solution I think what magnitization...- Lord33
- Thread
- Atom Magnetization
- Replies: 3
- Forum: Introductory Physics Homework Help
-
A
B Split an atom into each and every nucleon
Has anyone ever tried to split an atom up into each of its individual nucleon? If not, what's the closest anyone has come to? For example, if Uranium has 238 nucleons, has anyone ever tried to split it up into each and every last proton and neutron? Can anyone even do it for a light element like...- Albertgauss
- Thread
- Atom Nucleon Split
- Replies: 17
- Forum: High Energy, Nuclear, Particle Physics
-

Light absorption by a three level atom
Homework Statement We have 2 laser beams counter propagating through a cavity with an atom traveling at some velocity. So one laser beam is propagating left and the other right. One laser beam has an intensity of .05*I_sat and the other 10*I_sat. The goal is to get the transmittance (or...- BiGyElLoWhAt
- Thread
- Absorption Atom Light
- Replies: 3
- Forum: Engineering and Comp Sci Homework Help
-
L
I How many orbits are in the hydrogen atom?
How many orbits are in the hydrogen atom?- LSMOG
- Thread
- Atom Hydrogen Hydrogen atom Orbits
- Replies: 4
- Forum: Quantum Physics
-

I Exchange of photons inside atoms
I was reading Feynman Diagrams and stumbled upon this query: If the electrons and protons interact by exchange of photons, does the electron inside an atoms also interact with the nucleus with a similar kind of exchange?- CassiopeiaA
- Thread
- Atom Atoms Exchange Feynman diagram Photons
- Replies: 1
- Forum: Quantum Physics
-

Kinetic energy of the hydrogen atom in its ground state
I saw another post about this but i didn't quite find what i was looking for there so i thought i'd give it a go instead with a thread. Homework Statement Calculate the exact value of the kinetic energy of the hydrogen atom in its ground state. No more information is given, we are referred to...- Christofferk
- Thread
- Atom Energy Ground Ground state Hydrogen Hydrogen atom Kinetic Kinetic energy Physics Quantum State
- Replies: 22
- Forum: Advanced Physics Homework Help
-
C
I Location of donor atom electrons in N-type Silicon
I'm taking a module in solid state electronics and I'm a bit confused with the energy band diagrams. I was told that the region between the top of VB and and the bottom of the CB is the "forbidden gap". However after learning about N-type semiconductors(silicon), I see that the extra electron...- CoolDude420
- Thread
- Atom Electrons Silicon Solid state
- Replies: 3
- Forum: Atomic and Condensed Matter
-
E
E radial equation for a hydrogenic atom
Homework Statement By substituting R(r)=r^2*e^(-r/a) into the radial equation for a hydrogenic atom, find the values of a, ℓ and E for which the function R(r).Homework Equations http://puu.sh/rOJQ3/8bcad54c45.png The Attempt at a Solution I took the first derivative of R(r), then multiplied...- ElectricEel1
- Thread
- Atom Radial
- Replies: 1
- Forum: Advanced Physics Homework Help
-
J
Hydrogen Atom: Wavefunction collapse after measurement of Lz
Homework Statement Suppose we have a wavefunction with n=4. If we measure the orbital angular momentum along the z-direction(no spin in this problem) and get 2*hbar then what are the possible values of the total angular momentum and what is the most general wavefunction after the measurement...- Joker93
- Thread
- Angular momentum Atom Collapse Hydrogen Hydrogen atom Measurement Quantum mechanics Wavefunction Wavefunction collapse
- Replies: 20
- Forum: Advanced Physics Homework Help
-
B
B Understanding Pure States in Quantum Mechanics: From Atoms to Electrons
I learned about pure state, mixed state, reduce density matrix, etc. from this now famous paper http://philsci-archive.pitt.edu/5439/1/Decoherence_Essay_arXiv_version.pdf (thanks to Bill) I'd like to know something. Atty said somewhere: "A pure state means that we have prepared many copies of...- bluecap
- Thread
- Atom Pure Pure state State
- Replies: 18
- Forum: Quantum Physics
-
D
Modern Physics question -- an atom ejecting a relativistic electron
Homework Statement . An atom at rest can undergo radioactive decay, ejecting an electron at a maximum speed of 0.5c. If the atom in a particle accelerator is observed to produce an electron traveling at 0.75c, at least how fast must the atom itself have been moving? Homework Equations u0 x...- Dan350
- Thread
- Atom Electron Modern physics Physics Radioactive decay Relativistic Velocity transformation
- Replies: 3
- Forum: Introductory Physics Homework Help
-
K
Atom Volume Calc: Find the Volume of a Lead Atom
Homework Statement Lead has a mass of 11.35 g per cubic centimeter of volume, and the mass of one of its atoms is 3.439×10-25 kg. If the atoms are spherical and tightly packed, what is the volume of an individual atom? ( m3) Homework Equations conversions The Attempt at a Solution For this...- Kpgabriel
- Thread
- Atom Lead Volume
- Replies: 16
- Forum: Introductory Physics Homework Help
-
N
B Is it possible to squash an atom?
What would happen to an atom or molecule if you put it in a vice and tightened the vice as tight as possible?- NewToThis
- Thread
- Atom
- Replies: 26
- Forum: Other Physics Topics
-
A
B What would happen if nothing was like it is
HI guys! I was thinking... What would happen if the atom were considered the smallest thing of the universe, or just that electrons, protons e neutrons were that? That would impact in semiconductors technologies? Or plasma? Or nanotech? How? Thanks in advance.- Arthur Lopes
- Thread
- Atom Nanotechnology Particles Semiconductor Technology
- Replies: 2
- Forum: Other Physics Topics
-
I
I Solve Schrodinger Equation for Electrons in Atom: Potential Used?
How would you solve the Schrodinger equation for an electron in an atom. What potential do you use? Thanks!- Isaac0427
- Thread
- Atom Electrons
- Replies: 52
- Forum: Quantum Physics
-
B
B Can you find a single entangled atom in a haystack.
If you have a pair of particles (perhaps atoms for example) that are entangled, and one of them is mixed in amongst a whole bunch of other atoms of the same species, yet not entangled; could you differentiate in some way the entangled atom from the rest WITHOUT destroying the existing state of...- BernieM
- Thread
- Atom Entangled Entanglement
- Replies: 5
- Forum: Quantum Physics
-

Predict the hybridization and geometry of the indicated atom
Homework Statement Predict the hybridization and geometry of the indicated atom. Answer all parts. Homework Equations CH3CH2 There is a ( - ) sign above the second C Hybridization = ? Geometry = ? The Attempt at a Solution I know I'm supposed to count the groups around the atom, but I'm...- CMATT
- Thread
- Atom Geometry Hybridization
- Replies: 2
- Forum: Biology and Chemistry Homework Help
-
G
What is the Orbit of Hydrogen Atom for an Electron at 734 km/s?
Homework Statement On which orbit of hydrogen atom an electron has the speed of 734 km/s? Homework Equations Bohr's second postulate: mvr=nh,m=9.109\cdot 10^{-31}kg,v=734 km/s,h=6.626\cdot 10^{-34} m^{2}kg/s The Attempt at a Solution By using the second Bohr's postulate, we get 6686.006\cdot...- gruba
- Thread
- Atom Bohr model Bohr radius Electron Hydrogen Hydrogen atom Orbit
- Replies: 3
- Forum: Introductory Physics Homework Help
-
G
I Space Group Duplicates: Is it Possible for Monoatomic Crystals?
Hello, I was building some test crystal structures with VESTA software and I noticed when generating the POSCAR output files to be read by VASP that for a simple monoatomic basis such as in elemental silicon crystals some distinct space groups produce the same exact POSCAR files (i.e. same...- g_mogni
- Thread
- Atom Crystal Group Space Structure
- Replies: 5
- Forum: Atomic and Condensed Matter
-

I Visualizations of a hydrogen atom in an external electric field
I'd like to see some 3D visualizations of what the wave functions of the electron and proton in a hydrogen atom would look like in different applied electric fields. Say, have a reference image at 0V, then images at various voltages where the wave functions look interesting or just illustrative...- The Bill
- Thread
- Atom Electric Electric field Field Hydrogen Hydrogen atom
- Replies: 11
- Forum: Quantum Physics
-
D
Angular velocity of an atom in a magnetic field
Homework Statement The orbit of an electron (-e) around a nucleus (Ze) is a circular orbit of radius a in a plane perpendicular to a uniform magnetic field \textbf{B}. By writing the equation of motion in a frame rotating with the electron, show that the angular velocity \omega is given by one...- Dazed&Confused
- Thread
- Angular Angular velocity Atom Classical mechanics Field Magnetic Magnetic field Velocity
- Replies: 3
- Forum: Introductory Physics Homework Help
-
E
A The unit of atom scattering factor
Hi, I read in some papers which say the unit of atom scattering factor is e/atom. what is the meaning of that? thank you!- einstein1921
- Thread
- Atom Scattering Unit
- Replies: 1
- Forum: Atomic and Condensed Matter
-
R
B How were the first atoms created?
The Dalton's atomic theory states that atoms can neither be created nor destroyed. Then how was the first even atom created in this blank space? Is it physically possible to create an atom?- Ravyan786Asro
- Thread
- Atom Creation Universe
- Replies: 3
- Forum: Other Physics Topics
-
K
I Help to understand the wave function for atom (gas)
Hi there, I took the course of quantum mechanics long time ago. From there I learn how to describe an atom with wave function. For example, Hydrogen has the wave function in (spherical coordinates) space. In the book they consider a reduced mass for the nucleus and the only external electron...- KFC
- Thread
- Atom Function Gas Wave Wave function
- Replies: 5
- Forum: Quantum Physics
-

B Binding energy of the electron in Hydrogen atom.
This website here says that the expression for binding energy for an electron is: This http://ocw.mit.edu/high-school/chemistry/exam-prep/structure-of-matter/atomic-theory-and-atomic-structure/MITHFH_lecnotes05.pdfby MIT calculates it quantum mechanically to give: The book I was reading...- otaKu
- Thread
- Atom Binding energy Electron Energy Hydrogen Hydrogen atom
- Replies: 11
- Forum: Atomic and Condensed Matter
-
K
Electrostatic Energy in the Hydrogen Atom
Homework Statement We model the Hydrogen atom as a charge distribution in which the proton (a point charge) is surrounded by negative charge with the volume density of ρ = -ρ0 * exp (-2r/a0) where a0 is the Bohr radius. And ρ0 is a constant chosen such that the entire atomic distribution is...- Keiner Nichts
- Thread
- Atom Bohr model Electrostatic Electrostatic energy Electrostatics Energy Hydrogen Hydrogen atom
- Replies: 4
- Forum: Advanced Physics Homework Help
-
E
Can a strong electric field ionize an atom?
Metals have a work function which is the minimum energy needed to ionize an electron. I assume that the energy need is simply the energy difference of the two highest energy levels of a particular metal. This is all well and good and can be accomplished by absorbing a photon of that specific...- Electric to be
- Thread
- Atom Electric Electric field Field
- Replies: 1
- Forum: Electromagnetism
-

Does an Atom's Type dictate the Protons and Neutrons?
Hi, Does an Atom's Type have to do with the number of protons and neutrons it has? Or are they completely unrelated? Just curious. Thanks :) -
M
Solid State Physics - Modes of Oscillation of Atom
Homework Statement [/B] My question is more about the math of the problem. For equation 22.97, how do I construct a 3x3 matrix from a 12 element series? After constructing the matrix should I set the determinant equal to 0 where I will find the determinant has 3 solutions (2 being...- MightyDogg
- Thread
- Atom Modes Oscillation Physics Solid Solid state Solid state physics State
- Replies: 1
- Forum: Advanced Physics Homework Help
-
S
Why doesn't N3- have a dipole moment arrow from the central atom?
My teacher posted on his website, Lewis structures of different compounds. One of them were N3- Why isn't there a dipole moment arrow pointing from the central atom to the other Nitrogen? Does the arrow only point from neutral atom to negatively charged atom? -
N
Atom charge after ionizing (nuclei/particle) radiation
Homework Statement When an instable atom emits alpha or beta particles. Doesn't the overal electrical charge of the remaining atom change? For example when an atom emits alpha decay, a helium core is emitted, this results in the lose of two protons (forthe radiating atom). But doens't this mean...- n124122
- Thread
- Atom Charge Decay Electrical charge Ion Particle Radiation
- Replies: 2
- Forum: Introductory Physics Homework Help
-
G
I What is the Bremsstrahlung Spectrum function?
What is the function that describes the shape of this Bremsstrahlung X-ray spectrum curve? Ignore the characteristic "spikes".- greswd
- Thread
- Atom Bremsstrahlung Function Spectrum
- Replies: 2
- Forum: Atomic and Condensed Matter
-
M
I Hydrogen Atom Photon Emission Wavelength Formula
I am trying to calculate the Lyman-alpha wavelengths of photons emitted from different hydrogen-like atoms such as deuterium and positive helium ion 4He+, using the relation 1/λ = R*|1/ni^2 - 1/nf^2|, where R is the Rydberg constant and ni and nf are integer numbers corresponding to the initial...- math_major_111
- Thread
- Atom Emission Formula Hydrogen Hydrogen atom Lyman alpha Photon Photon emission Wavelength
- Replies: 11
- Forum: Quantum Physics
-
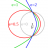
I Resonant Frequency of an Atom in an Atomic Clock
Beam standard atomic clocks work by changing the magnetic state of a cesium atom by applying a microwave frequency that matches the resonant frequency of a cesium atom. What is this resonant frequency of the atom in terms of actual events that occur on the atomic level...- Tom MS
- Thread
- Atom Atomic Atomic clock Clock Frequency Magnetic Resonance Resonant Resonant frequency
- Replies: 1
- Forum: Atomic and Condensed Matter
-
P
Determining <r> for the hydrogen atom
Homework Statement The equation for the normalized ##n=3##, ##l=2##, ##m=0## wavefunction is given by $$\psi_{320}=\frac{1}{81\sqrt{6\pi}}\left(\frac{1}{a_0}\right)^{3/2}\left(\frac{1}{a_0^2}\right)r^2e^{-\frac{r}{3a_0}}(3cos^2\theta-1)e^{i\phi}$$ Determine the expectation value ##<r>##...- Potatochip911
- Thread
- Atom Hydrogen Hydrogen atom
- Replies: 2
- Forum: Introductory Physics Homework Help
-
K
What is the correction to the wavelength of an emitted photon due to recoil?
Homework Statement When a photon is emitted by an atom, the atom must recoil to conserve momentum. This means that the photon and the recoiling atom share the transition energy. (a) For an atom with mass m, calculate the correction Δλ due to recoil to the wavelength of an emitted photon. Let λ...- kent davidge
- Thread
- Atom Speed
- Replies: 4
- Forum: Introductory Physics Homework Help
-

Difference between Rutherford and Bohr's theory of atom
Homework Statement The difference between Rutherford's and Bohr's theory of atom is ... A. Bohr's spectrum of hydrogen atom is discrete while Rutherford's continuous B. Bohr's stated electron goes around the nucleon while radiating energy, while Rutherford's not radiating energy. C. Bohr's...- terryds
- Thread
- Atom Difference Rutherford Theory
- Replies: 4
- Forum: Introductory Physics Homework Help
-
J
Effective Nuclear Charge for Sodium Atom
Homework Statement The 3s state Na has an energy of -5.14 eV. Determine the effective nuclear charge. Homework Equations ##Z_{eff} = Z - s## Where Z is the atomic number and s is the number of shells in inner orbits. The Attempt at a Solution The textbook doesn't go into much more detail...- James Brady
- Thread
- Atom Charge Nuclear Sodium
- Replies: 1
- Forum: Advanced Physics Homework Help
-
J
Angular momentum of hydrogen atom with Schrodinger Equation
If we were to assume that the electron moves around the proton with radius a, the Schrodinger equation becomes: ##\frac{1}{a^2}\frac{d^2\psi}{d\phi^2} + \frac{2m}{\hbar^2}|E|\psi = 0## The question in my textbook asks me to solve the above equation to obtain values of energy and angular...- James Brady
- Thread
- Angular Angular momentum Atom Bohr model Hydrogen Hydrogen atom Momentum Quantum mechanics Schrödinger Schrodinger equation
- Replies: 8
- Forum: Advanced Physics Homework Help